Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 I transfer models using experimental data of the "Plavsk scenario"
I transfer models using experimental data of the "Plavsk scenario"Zvonova, I.*; Krajewski, P.*; Berkovskyy, V.*; Ammann, M.*; Duffa, C.*; Filistovic, V.*; Homma, Toshimitsu; Kany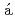 r, B.*; Nedveckaite, T.*; Simon, S.*; et al.
r, B.*; Nedveckaite, T.*; Simon, S.*; et al.
Proceedings of 7th International Conference on Nuclear and Radiochemistry (NRC-7) (CD-ROM), 5 Pages, 2008/08
Nagame, Yuichiro; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Akiyama, Kazuhiko; Ishii, Yasuo; Sato, Tetsuya; Hirata, Masaru; Nishinaka, Ichiro; Ichikawa, Shinichi; et al.
Radiochimica Acta, 93(9-10), p.519 - 526, 2005/00
Times Cited Count:31 Percentile:87.03(Chemistry, Inorganic & Nuclear)no abstracts in English
Xia, X.; Shibata, Masahiro; Kitamura, Akira; Kamei, Gento
P.779-781, p.779 - 781, 2004/08
A systematic sorption study on cesium, predicted to be a key radionuclide in the current safety assessment, was conducted on the sedimentary rock from the Horonobe URL, as the first step of linking sorption study in laboratory and modeling prediction of sorption at site.
Haba, Hiromitsu; Tsukada, Kazuaki; Asai, Masato; Nishinaka, Ichiro; Sakama, Minoru*; Goto, Shinichi*; Hirata, Masaru; Ichikawa, Shinichi; Nagame, Yuichiro; Kaneko, Tetsuya*; et al.
Radiochimica Acta, 89(11-12), p.733 - 736, 2002/02
Times Cited Count:15 Percentile:68.91(Chemistry, Inorganic & Nuclear)no abstracts in English
Nagame, Yuichiro; Zhao, Y.*; Nishinaka, Ichiro; Goto, Shinichi*; Kaji, D.*; Tanikawa, Masashi*; Tsukada, Kazuaki; Asai, Masato; Haba, Hiromitsu; Sakama, Minoru*; et al.
Radiochimica Acta, 89(11-12), p.681 - 688, 2002/02
Times Cited Count:3 Percentile:27.07(Chemistry, Inorganic & Nuclear)no abstracts in English
Watanabe, Satoshi; Ishioka, Noriko; Osa, Akihiko; Koizumi, Mitsuo; Sekine, Toshiaki; Kiyomiya, Shoichiro*; Nakanishi, Hiromi*; Mori, Satoshi*
Radiochimica Acta, 89(11-12), p.853 - 858, 2002/02
Times Cited Count:17 Percentile:74.75(Chemistry, Inorganic & Nuclear)no abstracts in English
Usuda, Shigekazu; Yasuda, Kenichiro
Proceedings of 5th International Conference on Nuclear and Radiochemistry (NRC5), 2, p.595 - 598, 2000/00
no abstracts in English
; McKinl, I. G.*; Mori, A.*; Frieg, B.*; W.Kickmaie*
Proceedings of International Conference on Nuclear and Radiochemistry, 0 Pages, 2000/00
None
Nagame, Yuichiro; Akiyama, Kazuhiko*; Asai, Masato; Goto, Shinichi*; Haba, Hiromitsu*; Ishii, Yasuo; Kasamatsu, Yoshitaka; Nishinaka, Ichiro; Sato, Tetsuya; Tome, Hayato*; et al.
no journal, ,
We present a study of fluoro complex formation of the transactinide elements, rutherfordium (Rf) and dubnium (Db), at JAEA (Japan Atomic Energy Agency). The transactinide nuclides  Rf and
Rf and  Db were produced in the reactions
Db were produced in the reactions  Cm(
Cm( O,5n) and
O,5n) and  Cm(
Cm( F,5n), respectively, at the JAEA tandem accelerator. Ion-exchange behavior of Rf and Db together with their lighter homologues in HF/HNO
F,5n), respectively, at the JAEA tandem accelerator. Ion-exchange behavior of Rf and Db together with their lighter homologues in HF/HNO mixed solutions has been investigated with a rapid ion-exchange separation apparatus AIDA. It has been found that fluoro complexation of Rf is significantly different from that of the homologues. A large difference in the adsorption behavior of Db and the homologue Ta on the anion-exchange resin has been also observed.
mixed solutions has been investigated with a rapid ion-exchange separation apparatus AIDA. It has been found that fluoro complexation of Rf is significantly different from that of the homologues. A large difference in the adsorption behavior of Db and the homologue Ta on the anion-exchange resin has been also observed.
Miyamoto, Yutaka; Yasuda, Kenichiro; Magara, Masaaki; Kimura, Takaumi
no journal, ,
A simple automatic system for sequential separation of U, Th, Pb, and the lanthanides using a single anion-exchange column was developed. The system consists of a tiny anion-exchange column, switching valves controlled by a PC, and a gas bottle to pressurize the eluents. The separation of elements of interest was optimized for several parameters including particle size, column length, and flow rate of eluents. The elements of interest were completely separated within 6 hours by use of this system.
 -ray activation analysis using pulsed neutron at J-PARC/ANNRI
-ray activation analysis using pulsed neutron at J-PARC/ANNRIToh, Yosuke; Harada, Hideo; Kimura, Atsushi; Nakamura, Shoji; Furutaka, Kazuyoshi; Kitatani, Fumito; Hara, Kaoru; Koizumi, Mitsuo; Ebihara, Mitsuru*
no journal, ,
The Accurate Neutron-Nucleus Reaction Measurement Instrument (ANNRI) has been constructed at the beamline No.04 at the Material and Life Science Experimental Facility (MLF) of the Japan Proton Accelerator Research Complex (J-PARC). ANNRI has been used for the measurement of nuclear cross-section data, nuclear astrophysics and prompt  -ray analysis (PGA). We have developed a time-of-flight prompt
-ray analysis (PGA). We have developed a time-of-flight prompt  -ray analysis combined with
-ray analysis combined with  -ray coincidence technique. This method will be improved a signal to noise ratio. Preliminary experiments of standard reference materials were made in ANNRI. In this study, the background spectra, the data acquisition dead time and the effect of the self-shield of neutron flux have been measured and evaluated.
-ray coincidence technique. This method will be improved a signal to noise ratio. Preliminary experiments of standard reference materials were made in ANNRI. In this study, the background spectra, the data acquisition dead time and the effect of the self-shield of neutron flux have been measured and evaluated.
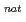 Pb reaction
Pb reactionNishinaka, Ichiro; Yokoyama, Akihiko*; Washiyama, Koshin*; Amano, Ryohei*; Maeda, Eita*; Yamada, Norihiro*; Makii, Hiroyuki; Watanabe, Shigeki; Ishioka, Noriko; Hashimoto, Kazuyuki
no journal, ,
Toyoshima, Atsushi; Oe, Kazuhiro; Asai, Masato; Miyashita, Sunao; Sato, Tetsuya; Sato, Nozomi; Kaneya, Yusuke*; Kitatsuji, Yoshihiro; Tsukada, Kazuaki; Nagame, Yuichiro; et al.
no journal, ,
Yokokita, Takuya*; Oe, Kazuhiro; Komori, Yukiko*; Kikutani, Yuki*; Kino, Aiko*; Nakamura, Kohei*; Kasamatsu, Yoshitaka*; Takahashi, Naruto*; Yoshimura, Takashi*; Takamiya, Koichi*; et al.
no journal, ,
We carried out solvent extraction of Mo(VI), Mo(V), W(VI) and W(V) in 0.01-0.36 M Aliquat 336 / 0.1-11 M HCl system as model experiments for element 106, seaborgium (Sg). The HCl solutions of Mo(VI), Mo(V), W(VI) and W(V) were prepared using macro amounts of Mo and W. Extraction behaviors of mononuclear Mo and W were also investigated using carrier-free radiotracers  Mo and
Mo and  W, which were produced as
W, which were produced as  U(n, f) and
U(n, f) and  Ta (p, n) reaction, respectively. The distribution ratios (D) of Mo(V) and W(V) were higher than those of Mo(VI) and W(VI), respectively. These results suggest that reduction behavior of the group-6 elements can be observed by solvent extraction. The D values of carrier-free Mo(VI) and W(VI) are almost the same as those with macro amounts in 6-11 M HCl. This condition would be suitable for the Sg experiments.
Ta (p, n) reaction, respectively. The distribution ratios (D) of Mo(V) and W(V) were higher than those of Mo(VI) and W(VI), respectively. These results suggest that reduction behavior of the group-6 elements can be observed by solvent extraction. The D values of carrier-free Mo(VI) and W(VI) are almost the same as those with macro amounts in 6-11 M HCl. This condition would be suitable for the Sg experiments.
Shinonaga, Taeko*; Markus, L.*; Okura, Takehisa
no journal, ,
The isotopic composition of uranium (U) in aerosol samples collected before and after the accident of the Fukushima Daiichi Nuclear Power Plant (FDI-NPP), occurred on March 11, 2011, was studied. The aerosol samples were collected on filters by an air sampling system in Tokai, Japan (at 120 km south-southwestern of the FDI-NPP). The U isotopic composition in each filter was analyzed by sector field inductively coupled plasma mass spectrometry (SF-ICPMS). The compositions of U in the samples collected after the accident show that non-natural U existed in the aerosol. The results of series measurements of radioactive materials before and after the accident indicate that the radioactive materials released from the FDI-NPP into the atmosphere were transported to the sampling station in Tokai, a place at 120 km distance from the FDI-NPP, within a day.
Omtvedt, J. P.*; Oe, Kazuhiro; Lerum, H. V.*; Toyoshima, Atsushi; Haba, Hiromitsu*; Tsukada, Kazuaki; Kratz, J. V.*; Nagame, Yuichiro; Sch del, M.
del, M.
no journal, ,
We are planning to investigate the redox potentials of element 106, seaborgium, with a combination of the automated liquid-liquid extraction system SISAK and a flow electrolytic column. In order to combine these two apparatuses, we need to reduce the liquid flow-rate of the SISAK system. In this study, a new degasser, which works with a lower flow rate, was developed for SISAK. It separates the gas-liquid mixture using a hydrophobic Teflon membrane (only the gas can go through the membrane). Using this new degasser, dissolution efficiencies of gas-jet transported products were measured. A high yield of around 80% was observed at a flow rate of 0.1 mL/s with a mixer for the gas-liquid mixing. This result has never been achieved with the conventional SISAK degasser. This is an important step forward with respect to combining the SISAK system with a flow electrolytic column for performing redox experiments on Sg.
Ohashi, Yusuke
no journal, ,
In this study we estimated the applicability of process using BMICl and chorin chloride-urea ionic liquid (CCU) for metal waste and NaF waste contaminated with uranium. Dissolution experiments were carried out by adding UF samples in BMICl and CCU in air. UF
samples in BMICl and CCU in air. UF samples were completely dissolved in BMICl after around 6h at 100
samples were completely dissolved in BMICl after around 6h at 100 C. 38% samples were also dissolved in CCU after 5h at 100
C. 38% samples were also dissolved in CCU after 5h at 100 C. 86% of uranium in Sodium fluoride (NaF) waste was also dissolved in BMICl after 3h at 100
C. 86% of uranium in Sodium fluoride (NaF) waste was also dissolved in BMICl after 3h at 100 C in air. 64% of uranium in NaF also dissolved in CCU after 3h at 100
C in air. 64% of uranium in NaF also dissolved in CCU after 3h at 100 C. Cyclic voltammograms of the sample solutions prepared by dissolving steel waste into BMICl were measured. The result suggests that electrolytic deposit of iron could not get mixed in with uranium deposit.
C. Cyclic voltammograms of the sample solutions prepared by dissolving steel waste into BMICl were measured. The result suggests that electrolytic deposit of iron could not get mixed in with uranium deposit.
Toh, Yosuke; Ebihara, Mitsuru*; Huang, M.; Kimura, Atsushi; Nakamura, Shoji; Harada, Hideo
no journal, ,
Asai, Shiho; Hanzawa, Yukiko; Konda, Miki; Suzuki, Daisuke; Magara, Masaaki; Kimura, Takaumi
no journal, ,
Isotopes of Zr and Mo including both radio and stable isotopes can be found in spent nuclear fuel. Of these isotopes,  Zr and
Zr and  Mo which have long half-lives of 1.61
Mo which have long half-lives of 1.61  10
10 y and 4
y and 4  10
10 y, respectively, are of great importance from the viewpoint of managing high-level radioactive wastes in a long-term basis. In this study, contents of Zr and Mo isotopes in spent nuclear fuel solution were determined by isotope dilution inductively coupled plasma mass spectrometry. The sample solution was prepared by dissolving a Japanese PWR irradiated UO
y, respectively, are of great importance from the viewpoint of managing high-level radioactive wastes in a long-term basis. In this study, contents of Zr and Mo isotopes in spent nuclear fuel solution were determined by isotope dilution inductively coupled plasma mass spectrometry. The sample solution was prepared by dissolving a Japanese PWR irradiated UO fuel with a burnup of 51 GWd/t. The measured contents of
fuel with a burnup of 51 GWd/t. The measured contents of  Zr,
Zr, 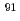 Zr,
Zr, 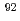 Zr,
Zr,  Zr,
Zr, 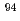 Zr, and
Zr, and 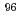 Zr in the sample were agreed well with the predicted values obtained through a burnup calculation code ORIGEN2. For Mo, the contents in the sample were approximately 30% less than the predicted values, indicating that part of Mo exists in the insoluble residue.
Zr in the sample were agreed well with the predicted values obtained through a burnup calculation code ORIGEN2. For Mo, the contents in the sample were approximately 30% less than the predicted values, indicating that part of Mo exists in the insoluble residue.